Arpid morgen mit Fundmentalentscheidung
19.11.08 19:32
#1
Heffalump
Arpid morgen mit Fundmentalentscheidung
Zulassung oder nicht. Da sind wir dabei.
Ist hier jemand investiert?
Ist hier jemand investiert?
19.11.08 19:39
#3
Heffalump
Um was gehts? Darum
PIVOTAL PHASE III DATA CONFIRM THE EFFICACY AND SAFETY OF ARPIDA’S INTRAVENOUS ICLAPRIM IN TREATING PATIENTS WITH COMPLICATED SKIN AND SKIN STRUCTURE INFECTIONS
Iclaprim also achieved high eradication rates for the most common pathogens, including methicillin-resistant Staphylococcus aureus (MRSA)
Reinach, Switzerland, 26 October 2008 – Arpida (SWX: ARPN) today presented the combined results from two pivotal Phase III clinical trials at the 48th Interscience Conference on Antimicrobial Agents and Chemotherapy (ICAAC)/Infectious Diseases Society of America (IDSA) 46th annual meeting in Washington, DC. In these studies, intravenous iclaprim, a novel antibiotic, showed high clinical cure rates which were similar to those of the comparator drug, linezolid, in the treatment of complicated skin and skin structure infections (cSSSI) caused by Gram-positive bacteria, including methicillin-resistant Staphylococcus aureus (MRSA). In addition, iclaprim was well-tolerated with a safety profile that compared favorably to linezolid. A New Drug Application for intravenous iclaprim in cSSSI will be discussed at the meeting of the U.S. Food and Drug Administration Anti-Infective Drugs Advisory Committee on 20 November 2008.
Iclaprim, a novel antibiotic from the trusted dihydrofolate reductase (DHFR) selective inhibitor class of antibiotics, was specifically designed to address the growing need for additional treatment options to combat resistant infections including MRSA. MRSA affects more than 2 million people in the United States each year and rates of hospital- and community-acquired MRSA are on the rise. [i, ii]
“The high efficacy rates and favorable tolerability profile of iclaprim strongly supports its potential as a new treatment option for patients who have acquired serious infections caused by MRSA,” said Dennis L. Stevens, MD, PhD, Veterans Affairs Medical Center, Boise, Idaho. “With rates of MRSA increasing and effectiveness of some current antibiotics declining, it is important to identify effective alternative treatments for these infections.”
The results presented at the ICAAC/IDSA meeting were gathered from the ASSIST-1 and ASSIST-2, (Arpida’s Skin and Skin Structure Infection STudy) pivotal Phase III studies involving a total of 991 patients. In a combined efficacy analysis, the clinical cure rate at the test-of-cure visit was 82.2 percent for iclaprim versus 85.3 percent for linezolid in the intent-to-treat population and cure rates were 92.3 percent and 97.8 percent, respectively, in the per protocol population. Data from the studies also show that iclaprim exhibited a high eradication rate for MRSA (76.4 percent), which was comparable to that of linezolid (78.7 percent).
Overall, iclaprim was found to be safe and well-tolerated at a dose of 0.8 mg/kg in the Phase III ASSIST trials. Adverse events were found to be less frequent among patients treated with iclaprim as compared to linezolid. In earlier Phase I trials, iclaprim was shown to have a low propensity for interactions with other drugs.
“These data further support the safety and efficacy of iclaprim as a potential treatment against complicated skin and skin structure infections,” said Jürgen Raths MD, President and CEO of Arpida. “Arpida remains encouraged and optimistic about the potential of iclaprim for the treatment of MRSA and related infections, and looks forward to bringing iclaprim to market, providing a new treatment option for physicians.”
The ASSIST Trials
The ASSIST trials were randomized, multi-center, double-blind, Phase III studies designed to establish the efficacy and safety of iclaprim in the treatment of patients with cSSSI known or suspected to be caused by Gram-positive pathogens. The trials were of essentially identical design, making a combined analysis of iclaprim possible in a larger population. A 95 percent confidence interval was used to determine statistical significance of study data and the pre-specified non-inferiority margin (-12.5 percent) was met in all populations in both trials.
A total of 991 patients, 18 years old and over with cSSSIs were enrolled in the trials. Patients were treated for 10 to 14 days with either 0.8 mg/kg iclaprim or 600 mg linezolid, both administered intravenously twice daily and analyzed based on populations, including intent-to-treat (ITT), modified intent-to-treat (MITT), per protocol (PP) and modified clinical evaluable (MCE).
• The ITT population included all patients who received at least one dose of medication;
• The MITT population included all patients in the ITT population who had an infecting Gram-positive pathogen isolated at baseline;
• The PP population excludes all patients with any protocol violation; and
• The MCE population was the same as the PP population, but adding back clinically evaluable patients whose protocol violation was use of additional systemic or topical prohibited antibiotics or high-dose steroids registered as clinical failures.
The causative pathogen distribution in the study population was well-balanced between treatment groups with Staphylococcus aureus (S. aureus) as the predominant pathogen isolated at baseline (76.5 percent for iclaprim and 81.1 percent for linezolid), of which 40 percent of isolates were MRSA.
Patients were evaluated daily for the first four days and then every other day thereafter during the treatment period, at the end of therapy, at the test-of-cure (TOC) visit (7 to 14 days post treatment) and at a late follow-up visit 7 to14 days after the TOC visit. The primary endpoint was the comparative clinical cure rates of iclaprim and linezolid at the TOC visit in the ITT and PP populations.
The most commonly reported adverse events in the clinical studies were: gastrointestinal disorders (7.4 percent versus 10.4 percent for iclaprim and linezolid, respectively); general disorders and administration site conditions (4.2 percent versus 3.9 percent); nervous system disorders (4.8 percent versus 6.5 percent); and skin and subcutaneous tissue disorders (4.2 percent versus 4.5 percent).
About MRSA
Methicillin-resistant Staphylococcus aureus (MRSA) is a type of bacteria that is resistant to most commonly available antibiotics.[iii] MRSA infections occur most frequently among persons who have weakened immune systems in hospitals and healthcare facilities; however community-associated-MRSA infections, such as abscesses, boils, and other pus-filled lesions, are increasingly being diagnosed in healthy people who have not been recently hospitalized or have undergone medical procedure.iii, [iv] The most common antibiotic used to treat MRSA infections is vancomycin, but recent evidence suggests resistance to vancomycin is on the rise.[v, vi]
Approximately 2.3 million people in the United States acquire MRSA and 89.4 million people are colonized with S. aureus annually, according to the Centers for Disease Control and Prevention (CDC).[i, vii] An estimated 292,000 hospitalizations with a diagnosis of S. aureus infection occur annually in U.S. hospitals and, of these, approximately 126,000 hospitalizations are related to MRSA.[viii] MRSA is responsible for an average of 94,000 life-threatening infections and 18,650 deaths each year in the United States.[ix]
About Iclaprim
Iclaprim is an antibiotic currently in development for the treatment of serious infections requiring hospitalization caused by Gram-positive bacteria, including those caused by MRSA. Iclaprim was designed to meet a growing medical need for additional treatment options to combat resistant infections and is the first antibiotic in the dihydrofolate reductase (DHFR) selective inhibitor class to demonstrate efficacy against cSSSIs caused by MRSA. The DHFR class has been proven safe and effective in more than four decades of clinical use.
In March 2008, Arpida completed the U.S. filing of the New Drug Application for intravenous iclaprim for the treatment of cSSSIs. The U.S. Food and Drug Administration defined a Prescription Drug User Fee Act (PDUFA) goal date of January 16, 2009. In August 2008, Arpida announced acceptance of its Marketing Authorization Application (MAA) for intravenous iclaprim for the treatment of cSSSIs for review by the European Medicines Agency. Arpida has also filed a marketing application in Canada.
Iclaprim is being studied across a range of serious infections caused by Gram-positive bacteria, with ongoing studies in patients with cSSSIs and hospital-acquired pneumonia (HAP), ventilator-acquired pneumonia (VAP) or healthcare-associated pneumonia (HCAP). Arpida is also pursuing the development of an oral formulation of iclaprim to provide treatment continuity for the patient from the hospital to the patient’s home.
About Arpida
Arpida (SWX: ARPN) is a biopharmaceutical company headquartered in Reinach, Switzerland with operations in Switzerland and the United States. It focuses on the discovery, development and commercialization of novel drugs for the treatment of microbial infections. Arpida has a fully integrated platform for the discovery and development of drug candidates to address the increasing prevalence of resistance of bacteria, such as methicillin-resistant Staphylococcus aureus (MRSA), to existing antibiotic therapies. Arpida is currently developing an iclaprim oral formulation as a step-down therapy after intravenous therapy. Apart from the flagship iclaprim program, Arpida has an innovative antifungal treatment in Phase III clinical development as well as several earlier-stage programs (AR-709 and AR-2474).
This press release contains specific forward-looking statements, e.g. statements including terms like believe, assume, expect or similar expressions. Such forward-looking statements are subject to known and unknown risks, uncertainties and other factors which may result in a substantial divergence between the actual results, financial situation, development or performance of the company and those explicitly or implicitly presumed in these statements. Against the background of these uncertainties readers should not place undue reliance on forward-looking statements. The company assumes no responsibility to update forward-looking statements or to adapt them to future events or developments.
Media contacts:
Dr Jürgen Raths, President and CEO
Tel: + 41 61 417 96 60
Harry Welten, MBA, CFO and Senior Vice President
Tel: + 41 61 417 96 65
Paul Verbraeken, Head of Corporate Communications
Tel: + 41 61 417 96 83
________________________________________
i Kuehnert MJ et al. Journal of Infectious Diseases. 2006; 193: 172-9.
[ii] Loffler, C.A. MacDougall C. Expert Review of Anti-Infective Therapy 2007; 5: 961-81.
[iii] Centers for Disease Control and Prevention Web site. Healthcare-Associated Methicillin Resistant Staphylococcus aureus (HA-MRSA) Overview. Available at http://www.cdc.gov/ncidod/dhqp/ar_MRSA.html. Accessed October 22, 2008.
[iv] Centers for Disease Control and Prevention Web site. Community-Associated Methicillin Resistant Staphylococcus aureus (CA-MRSA) Overview. Available at http://www.cdc.gov/ncidod/dhqp/ar_mrsa_ca.html. Accessed October 22, 2008.
[v] Sakoulas, G et al. Clinical Infectious Diseases 2006; 42: S40-S50.
[vi] Tenover F, Moellering RC Jr. Clinical Infectious Diseases 2007; 44: 1208-15.
[vii] Centers for Disease Control and Prevention Web site. S. aureus and MRSA Surveillance Summary 2007. Available at http://www.cdc.gov/ncidod/dhqp/ar_mrsa_surveillanceFS.html. Accessed October 22, 2008.
[viii] Kuehnert MJ et al. Emerging Infectious Diseases 2005; 11: 868-72.
[ix] Klevens, RM et al. Journal of the American Medical Association. 2007; 298: 1763-71.
26.10.2008 | Paul Verbraeken
Iclaprim also achieved high eradication rates for the most common pathogens, including methicillin-resistant Staphylococcus aureus (MRSA)
Reinach, Switzerland, 26 October 2008 – Arpida (SWX: ARPN) today presented the combined results from two pivotal Phase III clinical trials at the 48th Interscience Conference on Antimicrobial Agents and Chemotherapy (ICAAC)/Infectious Diseases Society of America (IDSA) 46th annual meeting in Washington, DC. In these studies, intravenous iclaprim, a novel antibiotic, showed high clinical cure rates which were similar to those of the comparator drug, linezolid, in the treatment of complicated skin and skin structure infections (cSSSI) caused by Gram-positive bacteria, including methicillin-resistant Staphylococcus aureus (MRSA). In addition, iclaprim was well-tolerated with a safety profile that compared favorably to linezolid. A New Drug Application for intravenous iclaprim in cSSSI will be discussed at the meeting of the U.S. Food and Drug Administration Anti-Infective Drugs Advisory Committee on 20 November 2008.
Iclaprim, a novel antibiotic from the trusted dihydrofolate reductase (DHFR) selective inhibitor class of antibiotics, was specifically designed to address the growing need for additional treatment options to combat resistant infections including MRSA. MRSA affects more than 2 million people in the United States each year and rates of hospital- and community-acquired MRSA are on the rise. [i, ii]
“The high efficacy rates and favorable tolerability profile of iclaprim strongly supports its potential as a new treatment option for patients who have acquired serious infections caused by MRSA,” said Dennis L. Stevens, MD, PhD, Veterans Affairs Medical Center, Boise, Idaho. “With rates of MRSA increasing and effectiveness of some current antibiotics declining, it is important to identify effective alternative treatments for these infections.”
The results presented at the ICAAC/IDSA meeting were gathered from the ASSIST-1 and ASSIST-2, (Arpida’s Skin and Skin Structure Infection STudy) pivotal Phase III studies involving a total of 991 patients. In a combined efficacy analysis, the clinical cure rate at the test-of-cure visit was 82.2 percent for iclaprim versus 85.3 percent for linezolid in the intent-to-treat population and cure rates were 92.3 percent and 97.8 percent, respectively, in the per protocol population. Data from the studies also show that iclaprim exhibited a high eradication rate for MRSA (76.4 percent), which was comparable to that of linezolid (78.7 percent).
Overall, iclaprim was found to be safe and well-tolerated at a dose of 0.8 mg/kg in the Phase III ASSIST trials. Adverse events were found to be less frequent among patients treated with iclaprim as compared to linezolid. In earlier Phase I trials, iclaprim was shown to have a low propensity for interactions with other drugs.
“These data further support the safety and efficacy of iclaprim as a potential treatment against complicated skin and skin structure infections,” said Jürgen Raths MD, President and CEO of Arpida. “Arpida remains encouraged and optimistic about the potential of iclaprim for the treatment of MRSA and related infections, and looks forward to bringing iclaprim to market, providing a new treatment option for physicians.”
The ASSIST Trials
The ASSIST trials were randomized, multi-center, double-blind, Phase III studies designed to establish the efficacy and safety of iclaprim in the treatment of patients with cSSSI known or suspected to be caused by Gram-positive pathogens. The trials were of essentially identical design, making a combined analysis of iclaprim possible in a larger population. A 95 percent confidence interval was used to determine statistical significance of study data and the pre-specified non-inferiority margin (-12.5 percent) was met in all populations in both trials.
A total of 991 patients, 18 years old and over with cSSSIs were enrolled in the trials. Patients were treated for 10 to 14 days with either 0.8 mg/kg iclaprim or 600 mg linezolid, both administered intravenously twice daily and analyzed based on populations, including intent-to-treat (ITT), modified intent-to-treat (MITT), per protocol (PP) and modified clinical evaluable (MCE).
• The ITT population included all patients who received at least one dose of medication;
• The MITT population included all patients in the ITT population who had an infecting Gram-positive pathogen isolated at baseline;
• The PP population excludes all patients with any protocol violation; and
• The MCE population was the same as the PP population, but adding back clinically evaluable patients whose protocol violation was use of additional systemic or topical prohibited antibiotics or high-dose steroids registered as clinical failures.
The causative pathogen distribution in the study population was well-balanced between treatment groups with Staphylococcus aureus (S. aureus) as the predominant pathogen isolated at baseline (76.5 percent for iclaprim and 81.1 percent for linezolid), of which 40 percent of isolates were MRSA.
Patients were evaluated daily for the first four days and then every other day thereafter during the treatment period, at the end of therapy, at the test-of-cure (TOC) visit (7 to 14 days post treatment) and at a late follow-up visit 7 to14 days after the TOC visit. The primary endpoint was the comparative clinical cure rates of iclaprim and linezolid at the TOC visit in the ITT and PP populations.
The most commonly reported adverse events in the clinical studies were: gastrointestinal disorders (7.4 percent versus 10.4 percent for iclaprim and linezolid, respectively); general disorders and administration site conditions (4.2 percent versus 3.9 percent); nervous system disorders (4.8 percent versus 6.5 percent); and skin and subcutaneous tissue disorders (4.2 percent versus 4.5 percent).
About MRSA
Methicillin-resistant Staphylococcus aureus (MRSA) is a type of bacteria that is resistant to most commonly available antibiotics.[iii] MRSA infections occur most frequently among persons who have weakened immune systems in hospitals and healthcare facilities; however community-associated-MRSA infections, such as abscesses, boils, and other pus-filled lesions, are increasingly being diagnosed in healthy people who have not been recently hospitalized or have undergone medical procedure.iii, [iv] The most common antibiotic used to treat MRSA infections is vancomycin, but recent evidence suggests resistance to vancomycin is on the rise.[v, vi]
Approximately 2.3 million people in the United States acquire MRSA and 89.4 million people are colonized with S. aureus annually, according to the Centers for Disease Control and Prevention (CDC).[i, vii] An estimated 292,000 hospitalizations with a diagnosis of S. aureus infection occur annually in U.S. hospitals and, of these, approximately 126,000 hospitalizations are related to MRSA.[viii] MRSA is responsible for an average of 94,000 life-threatening infections and 18,650 deaths each year in the United States.[ix]
About Iclaprim
Iclaprim is an antibiotic currently in development for the treatment of serious infections requiring hospitalization caused by Gram-positive bacteria, including those caused by MRSA. Iclaprim was designed to meet a growing medical need for additional treatment options to combat resistant infections and is the first antibiotic in the dihydrofolate reductase (DHFR) selective inhibitor class to demonstrate efficacy against cSSSIs caused by MRSA. The DHFR class has been proven safe and effective in more than four decades of clinical use.
In March 2008, Arpida completed the U.S. filing of the New Drug Application for intravenous iclaprim for the treatment of cSSSIs. The U.S. Food and Drug Administration defined a Prescription Drug User Fee Act (PDUFA) goal date of January 16, 2009. In August 2008, Arpida announced acceptance of its Marketing Authorization Application (MAA) for intravenous iclaprim for the treatment of cSSSIs for review by the European Medicines Agency. Arpida has also filed a marketing application in Canada.
Iclaprim is being studied across a range of serious infections caused by Gram-positive bacteria, with ongoing studies in patients with cSSSIs and hospital-acquired pneumonia (HAP), ventilator-acquired pneumonia (VAP) or healthcare-associated pneumonia (HCAP). Arpida is also pursuing the development of an oral formulation of iclaprim to provide treatment continuity for the patient from the hospital to the patient’s home.
About Arpida
Arpida (SWX: ARPN) is a biopharmaceutical company headquartered in Reinach, Switzerland with operations in Switzerland and the United States. It focuses on the discovery, development and commercialization of novel drugs for the treatment of microbial infections. Arpida has a fully integrated platform for the discovery and development of drug candidates to address the increasing prevalence of resistance of bacteria, such as methicillin-resistant Staphylococcus aureus (MRSA), to existing antibiotic therapies. Arpida is currently developing an iclaprim oral formulation as a step-down therapy after intravenous therapy. Apart from the flagship iclaprim program, Arpida has an innovative antifungal treatment in Phase III clinical development as well as several earlier-stage programs (AR-709 and AR-2474).
This press release contains specific forward-looking statements, e.g. statements including terms like believe, assume, expect or similar expressions. Such forward-looking statements are subject to known and unknown risks, uncertainties and other factors which may result in a substantial divergence between the actual results, financial situation, development or performance of the company and those explicitly or implicitly presumed in these statements. Against the background of these uncertainties readers should not place undue reliance on forward-looking statements. The company assumes no responsibility to update forward-looking statements or to adapt them to future events or developments.
Media contacts:
Dr Jürgen Raths, President and CEO
Tel: + 41 61 417 96 60
Harry Welten, MBA, CFO and Senior Vice President
Tel: + 41 61 417 96 65
Paul Verbraeken, Head of Corporate Communications
Tel: + 41 61 417 96 83
________________________________________
i Kuehnert MJ et al. Journal of Infectious Diseases. 2006; 193: 172-9.
[ii] Loffler, C.A. MacDougall C. Expert Review of Anti-Infective Therapy 2007; 5: 961-81.
[iii] Centers for Disease Control and Prevention Web site. Healthcare-Associated Methicillin Resistant Staphylococcus aureus (HA-MRSA) Overview. Available at http://www.cdc.gov/ncidod/dhqp/ar_MRSA.html. Accessed October 22, 2008.
[iv] Centers for Disease Control and Prevention Web site. Community-Associated Methicillin Resistant Staphylococcus aureus (CA-MRSA) Overview. Available at http://www.cdc.gov/ncidod/dhqp/ar_mrsa_ca.html. Accessed October 22, 2008.
[v] Sakoulas, G et al. Clinical Infectious Diseases 2006; 42: S40-S50.
[vi] Tenover F, Moellering RC Jr. Clinical Infectious Diseases 2007; 44: 1208-15.
[vii] Centers for Disease Control and Prevention Web site. S. aureus and MRSA Surveillance Summary 2007. Available at http://www.cdc.gov/ncidod/dhqp/ar_mrsa_surveillanceFS.html. Accessed October 22, 2008.
[viii] Kuehnert MJ et al. Emerging Infectious Diseases 2005; 11: 868-72.
[ix] Klevens, RM et al. Journal of the American Medical Association. 2007; 298: 1763-71.
26.10.2008 | Paul Verbraeken
20.11.08 20:51
#6
sertralin19
:-(
20.11.2008 19:45
UPDATE 1-U.S. panel votes against Arpida antibiotic
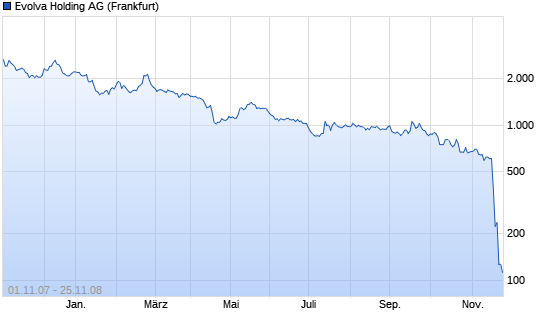
NEW YORK, Nov 20 (Reuters) - A U.S. regulatory panel has voted against recommending approval of Arpida's intravenous antibiotic iclaprim, the Swiss biotech company said on Thursday.
The Food and Drug Administration committee rejected the drug by 17 to two votes for the treatment of patients with complicated skin and skin structure infections.
The FDA, which is due to give a final decision on the medicine by Jan. 16, is not bound by the advisory committee's recommendations but usually draws heavily on its advice when making decisions.
Arpida (News) has limited cash as it does not yet have a product on the market and investors had been hoping for a swift approval for iclaprim, particularly given the difficulty of raising more funds in such tough markets.
Arpida's shares have plummeted this week, losing more than half of their value on concerns that the FDA would not approve iclaprim, the company's lead drug candidate.
UPDATE 1-U.S. panel votes against Arpida antibiotic
NEW YORK, Nov 20 (Reuters) - A U.S. regulatory panel has voted against recommending approval of Arpida's intravenous antibiotic iclaprim, the Swiss biotech company said on Thursday.
The Food and Drug Administration committee rejected the drug by 17 to two votes for the treatment of patients with complicated skin and skin structure infections.
The FDA, which is due to give a final decision on the medicine by Jan. 16, is not bound by the advisory committee's recommendations but usually draws heavily on its advice when making decisions.
Arpida (News) has limited cash as it does not yet have a product on the market and investors had been hoping for a swift approval for iclaprim, particularly given the difficulty of raising more funds in such tough markets.
Arpida's shares have plummeted this week, losing more than half of their value on concerns that the FDA would not approve iclaprim, the company's lead drug candidate.
21.11.08 09:20
#7
Heffalump
Interessantes BID ASK kurz vor dem Tod
Stuttgart 400 0,909 € 0,961 € 200 5,72% 09:00:04
Frankfurt 5.000 1,30 € 1,40 € 5.000 7,69% 08:59:05
Berlin 1.000 0,548 € 1,37 € 1.000 150,00% 08:57:46
|
Frankfurt als Fake?
Frankfurt 5.000 1,30 € 1,40 € 5.000 7,69% 08:59:05
Berlin 1.000 0,548 € 1,37 € 1.000 150,00% 08:57:46
|
Frankfurt als Fake?
26.01.10 10:06
#11
Heffalump
550% seit Eröffnung, und das mit den schlechten
news zu Beginn, was ein Joint Venture und/oder Übernahme bewirken kann..unglaublich
24.01.11 16:22
#14
chicken84
Evolva
Hey all,
Irgendwelche Einschätzungen zu Evolva. Die kurzfristigen Kurssprünge des letzten Jahres wirds heuer wohl nicht geben, aber ein Nachkauf um die CHF 1.50 kann wohl getrost ins Auge gefasst werden, oder?
Irgendwelche Einschätzungen zu Evolva. Die kurzfristigen Kurssprünge des letzten Jahres wirds heuer wohl nicht geben, aber ein Nachkauf um die CHF 1.50 kann wohl getrost ins Auge gefasst werden, oder?
15.02.11 16:09
#15
Bioshare
EVE in den Startlöchern!
Hallo Chicken84, Ich denke der Deal zwischen IFF und Evolva ist unterbewertet worden, eine Korrektur ist aber erst zu erwarten, wenn wir uns auf den 23. Mai zubewegen. Bin zuversichtlich für solide EVOLVA Kurse gegen 1.90 auf den Herbst hin. Falls du schon investiert bist, warte noch ab mit Nachkaufen, der Kurs wird vermutlich wegen der Analyse von Vontobel unter 1.40 tauchen. Danach heisst es hoffentlich Feuer frei! Das bisher gehandelte Volumen war zwar nicht glamurös aber jeder der rechnen kann sieht, dass sich EVE auf einem guten Weg befindet.
21.02.11 13:41
#16
Bioshare
Hoffe du liegst richtig
Das Volumen der letzten Tage gibt dir insofern Recht, als das dass Vertrauen wieder etwas zurückgekehrt ist.
10.09.13 10:26
#17
ruck
News vom 09.09.2103
Das sind sensationelle News - die Partnerschaft mit Cargrill zeigt bereits erste positive Ergebnisse. Die Aussichten dieser Kooperation sind meines Erachtens äußerst vielversprechend: http://www.pressebox.de/inaktiv/evolva-holding-sa/...ein/boxid/623496
10.09.13 11:44
#18
Ice-bein
# ruck
Das sind sensationelle News in der Tat. Das hat sich auch im gestrigen Kursverlauf wiedergespiegelt.
Was ich nicht verstehe, ist das sich der Kurs für Stevia First gestern nicht nach Norden bewegt hat. Schliesslich würden sie indirekt am Erfolg von Evolva durch Patentrechte profitieren.
Was ich nicht verstehe, ist das sich der Kurs für Stevia First gestern nicht nach Norden bewegt hat. Schliesslich würden sie indirekt am Erfolg von Evolva durch Patentrechte profitieren.
24.10.13 09:48
#20
stars123
3. Quartal
Weiss jemand wann die Quartalszahlen bekannt gegeben werden??
29.10.13 10:51
#22
stars123
...
Wie steht es um Evolva?? ist ja noch sehr viel luft nach oben??
zur zeit bewegt sich ja gar nichts.
29.11.13 12:33
#23
sertralin19
gar nichts?
also bei mir - ich halte evolva schon länger - gab es 250 % plus in 2013. dank nachkauf bei 35 ct macht sich das deutlich bemerkbar im depot. die aktie liegt in einem schönen aufwärtstrend. was soll sich denn noch bewegen?
lg
s19
lg
s19
31.01.14 14:59
#25
Ice-bein
Investing In Companies That Will Profit From The G
Investing In Companies That Will Profit From The Growing Healthier Food Market